Dr Rupam Tripura

Contact information
Research groups
Rupam Tripura
Clinical Researcher
Dr Rupam Tripura is a clinical researcher based at the Mahidol-Oxford Topical Medicine Research Unit (MORU) research stations in Cambodia. The main focus of his work has been to conduct clinical trials to understand the nature of drug-resistant falciparum malaria in western Cambodia and to develop treatment regimens, and strategies to combat the spread of resistance.
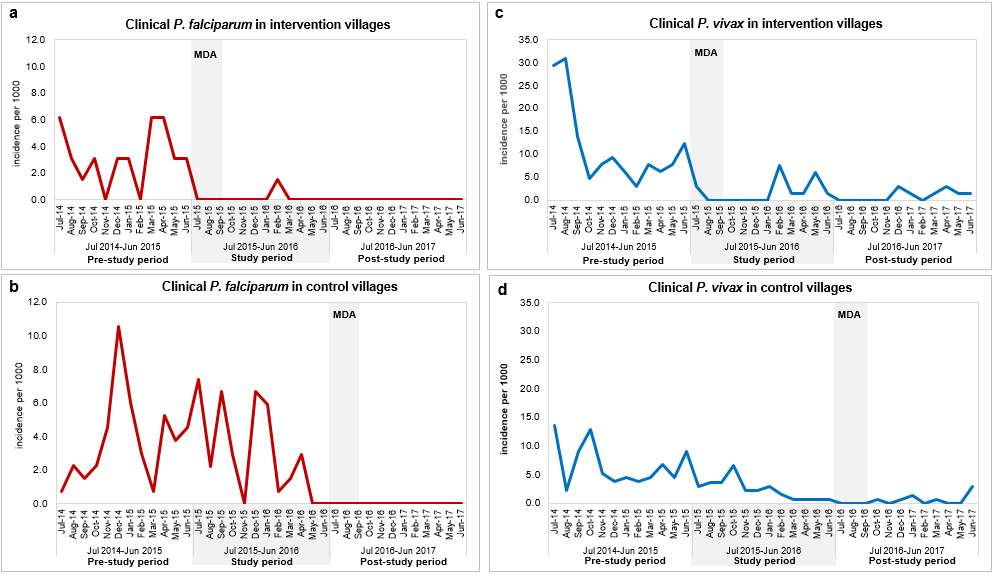
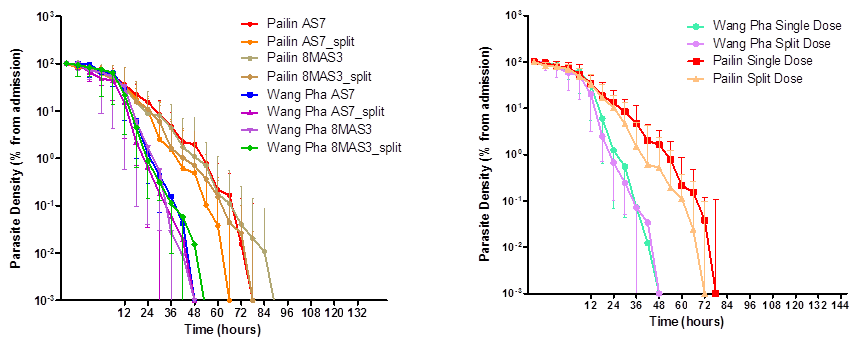
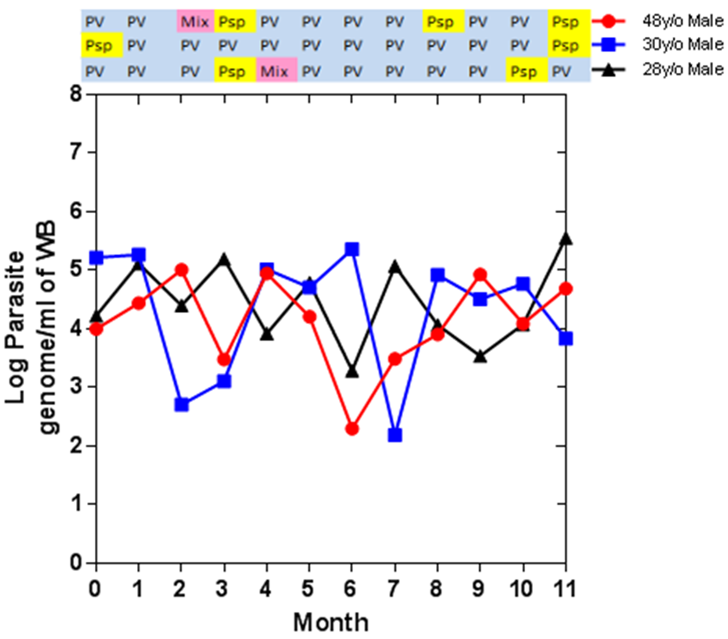
In 2008-2010, after the first reports of artemisinin resistant falciparum malaria, he was involved in a trial to look at the effect of high-dose or split-dose artesunate against artemisinin-resistant falciparum in western Cambodia. In 2011-13, he led the research team in Pailin, a site for the Tracking Resistance to Artemisinins Collaboration study (TRAC), an open-label, multicentre clinical trial conducted. In 2014-16, he was involved in the Triple Artemisinin Combination (TRAC 2) study in western Cambodia, which evaluated the efficacy, safety, and tolerability of combining three antimalarial treatments against standard artemisinin combination therapy (ACT). During 2013-14, He led a series of community based epidemiological studies to understand asymptomatic plasmodium infections and explore whether these are a barrier to malaria elimination. Therefore, during 2015-17, he jointly led the Cambodian sites for a multicentre clinical trial entitled Targeted Malaria Elimination (TME) which evaluated the effectiveness, safety and acceptability of three round of mass drug administration (MDA) with dihydroartemisinin-piperaquine in areas of multidrug resistant falciparum malaria. Currently, he is conducting in a clinical trial in Northern and Western part of Cambodia which evaluates artemether-lumefantrine with or without amodiaquine. The aim is to develop a treatment regimen against multi-drug resistant falciparum malaria.
Recent publications
Changing genetic profiles of Plasmodium falciparum piperaquine resistance in Southeast Asia over 25 years.
Journal article
Srisutham S. et al, (2026), Antimicrobial agents and chemotherapy, 70
Estimating enteric fever seroincidence in rural western Cambodia: findings from a population-based cross-sectional serosurvey
Journal article
Zhang M. et al, (2026)
Population pharmacokinetics of artemether–lumefantrine plus amodiaquine in patients with uncomplicated Plasmodium falciparum malaria
Journal article
Ding J. et al, (2026), British Journal of Clinical Pharmacology, 92, 589 - 605
Effectiveness and safety of 7-day high-dose primaquine and single-dose tafenoquine versus 14-day low-dose primaquine in patients with Plasmodium vivax malaria (EFFORT): a multicentre, open-label, randomised, controlled, superiority trial
Journal article
Degaga TS. et al, (2026), The Lancet Infectious Diseases
Moving towards high-dose primaquine or single-dose tafenoquine for Plasmodium vivax treatment in Cambodia: a meeting report from dissemination of results of the EFFORT trial to stakeholders
Journal article
Dysoley L. et al, (2026), Malaria Journal, 25